Single-Molecule Projects
Ultrasensitive Detection and Imaging of Molecules in Condensed Matter
Interferometric Image Scanning Microscopy (iISM) for label-free imaging at 120 nm lateral resolution inside live cells
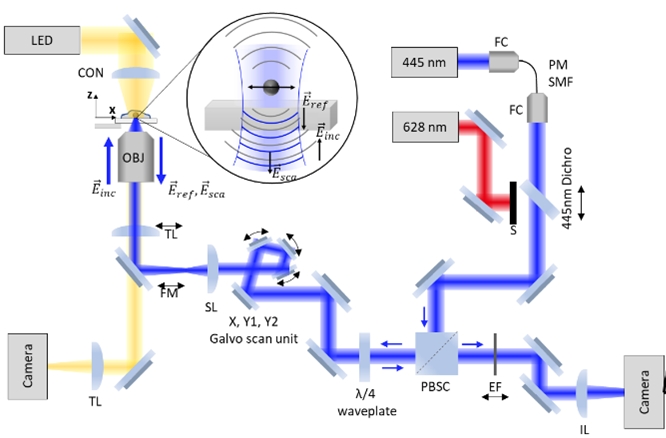 We have developed interferometric Image Scanning Microscopy (iISM) . By combining principles of confocal-iSCAT with those of Image Scanning Microscopy (ISM) , iISM pushes the boundaries of spatial resolution, contrast and sensitivity in label-free microscopy. The key idea is to replace the physical pinhole and point detector of C-iSCAT with an array detector, enabling acquisition of the full interferometric point-spread function (iPSF) at each scan position. Analogous to fluorescence ISM, the magnification is chosen such that each pixel on the array detector corresponds to an effectively closed pinhole confocal detection. The signals from the parallel acquired off-axis pinholes are reassigned to extract closed-pinhole lateral resolution while increasing the contrast-to-noise-ratio (CNR). To adapt the ISM concept to interferometric detection, we developed a modified adaptive pixel-reassignment algorithm that accounts for the encoded phase of the iPSF prior to the reassignment. By developing and implementing the first interferometric ISM microscope and by optimizing the iPSF, we achieve ~120 nm lateral resolution while exceeding the CNR of an open-pinhole confocal microscope, enabling either ~10× faster imaging at the same laser power or a ~10-fold reduction in illumination power at the same imaging speed, an important advantage for live-cell viability. Using iISM, we visualize intracellular organelles such as the endoplasmic reticulum, lysosomes, mitochondria, and vesicles label-free in live specimens over extended durations. Crucially, iISM is compatible with simultaneous confocal fluorescence microscopy, enabling correlative imaging that combines label-free structural (contextual) and dynamic information with molecular specificity when needed. This capability opens new avenues for quantitative studies of intracellular trafficking, hostpathogen interactions, cytoskeletal rearrangements, and organelle network dynamics under near-native conditions. iISM thus represents a powerful new tool to probe nanoscale live-cell dynamics with high resolution and minimal perturbation, paving the way for new biological insights. We have developed interferometric Image Scanning Microscopy (iISM) . By combining principles of confocal-iSCAT with those of Image Scanning Microscopy (ISM) , iISM pushes the boundaries of spatial resolution, contrast and sensitivity in label-free microscopy. The key idea is to replace the physical pinhole and point detector of C-iSCAT with an array detector, enabling acquisition of the full interferometric point-spread function (iPSF) at each scan position. Analogous to fluorescence ISM, the magnification is chosen such that each pixel on the array detector corresponds to an effectively closed pinhole confocal detection. The signals from the parallel acquired off-axis pinholes are reassigned to extract closed-pinhole lateral resolution while increasing the contrast-to-noise-ratio (CNR). To adapt the ISM concept to interferometric detection, we developed a modified adaptive pixel-reassignment algorithm that accounts for the encoded phase of the iPSF prior to the reassignment. By developing and implementing the first interferometric ISM microscope and by optimizing the iPSF, we achieve ~120 nm lateral resolution while exceeding the CNR of an open-pinhole confocal microscope, enabling either ~10× faster imaging at the same laser power or a ~10-fold reduction in illumination power at the same imaging speed, an important advantage for live-cell viability. Using iISM, we visualize intracellular organelles such as the endoplasmic reticulum, lysosomes, mitochondria, and vesicles label-free in live specimens over extended durations. Crucially, iISM is compatible with simultaneous confocal fluorescence microscopy, enabling correlative imaging that combines label-free structural (contextual) and dynamic information with molecular specificity when needed. This capability opens new avenues for quantitative studies of intracellular trafficking, hostpathogen interactions, cytoskeletal rearrangements, and organelle network dynamics under near-native conditions. iISM thus represents a powerful new tool to probe nanoscale live-cell dynamics with high resolution and minimal perturbation, paving the way for new biological insights.
Michelle Küppers and W.E. Moerner, Interferometric Image Scanning Microscopy for label-free imaging at 120 nm lateral resolution inside live cells, Light: Science & Applications 15, 129 (2026) (DOI: 10.1038/s41377-026-02210-y published online 27 Feb 2026). DOI
Stanford Report News Article
Stimulated emission does not radiate in a pure dipole pattern
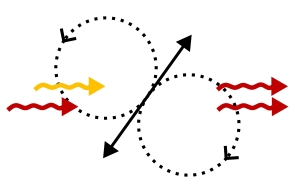 Stimulated Emission (StE) remains relatively unused as an image-forming signal, despite having potential advantages over fluorescence in speed, coherence, and ultimately resolution. Several ideas for the radiation pattern and directionality of StE remain prevalent, namely whether a single molecule would radiate StE itself in a pure dipole pattern, or whether its emission direction depends on the driving field. Previous StE imaging has been carried out in transmission, which would collect signal either way. Here, we introduce the StE driving field (the probe) at an angle, using total internal reflection to avoid incident probe light and its specular reflections in our detection path. In this non-collinear detection configuration which also collects some fluorescence from the sample, we observe fluorescence depletion even in the spectral window where an increase in detected signal from StE would be expected if StE radiated like a simple classical dipole. Because simultaneous direct measurement of the fluorescence represents a calibration of the potential size of StE were it spatially patterned like a classical dipole emitter, our study clarifies a critical characteristic of StE for optimal microscope design, optical cooling, and applications using small arrays of emitters. Stimulated Emission (StE) remains relatively unused as an image-forming signal, despite having potential advantages over fluorescence in speed, coherence, and ultimately resolution. Several ideas for the radiation pattern and directionality of StE remain prevalent, namely whether a single molecule would radiate StE itself in a pure dipole pattern, or whether its emission direction depends on the driving field. Previous StE imaging has been carried out in transmission, which would collect signal either way. Here, we introduce the StE driving field (the probe) at an angle, using total internal reflection to avoid incident probe light and its specular reflections in our detection path. In this non-collinear detection configuration which also collects some fluorescence from the sample, we observe fluorescence depletion even in the spectral window where an increase in detected signal from StE would be expected if StE radiated like a simple classical dipole. Because simultaneous direct measurement of the fluorescence represents a calibration of the potential size of StE were it spatially patterned like a classical dipole emitter, our study clarifies a critical characteristic of StE for optimal microscope design, optical cooling, and applications using small arrays of emitters.
Andrew E. S. Barentine, W. E. Moerner, Stimulated emission does not radiate in a pure dipole pattern, Optica 11, 464 (2024) (DOI: 10.1364/OPTICA.515226, published online 29 March 2024). DOI
Earlier Projects:
- Interferometry of a Single Nanoparticle Using the Gouy Phase of a Focused Laser Beam, Opt. Commun. 280, 487 (2007) [PDF]
- Scanning Interferometric Microscopy
for the Detection of Ultrasmall Phase Shifts in Condensed
Matter [Slides] [PDF]
- Exploring
Novel Methods of Interferometric Detection of Ultrasmall
Phase Shifts (Proc. SPIE v. 4962, 110 (2003)) [PDF]
- Pumping Single Molecules with Whispering
Gallery Modes of Microspheres [Abstract]
|